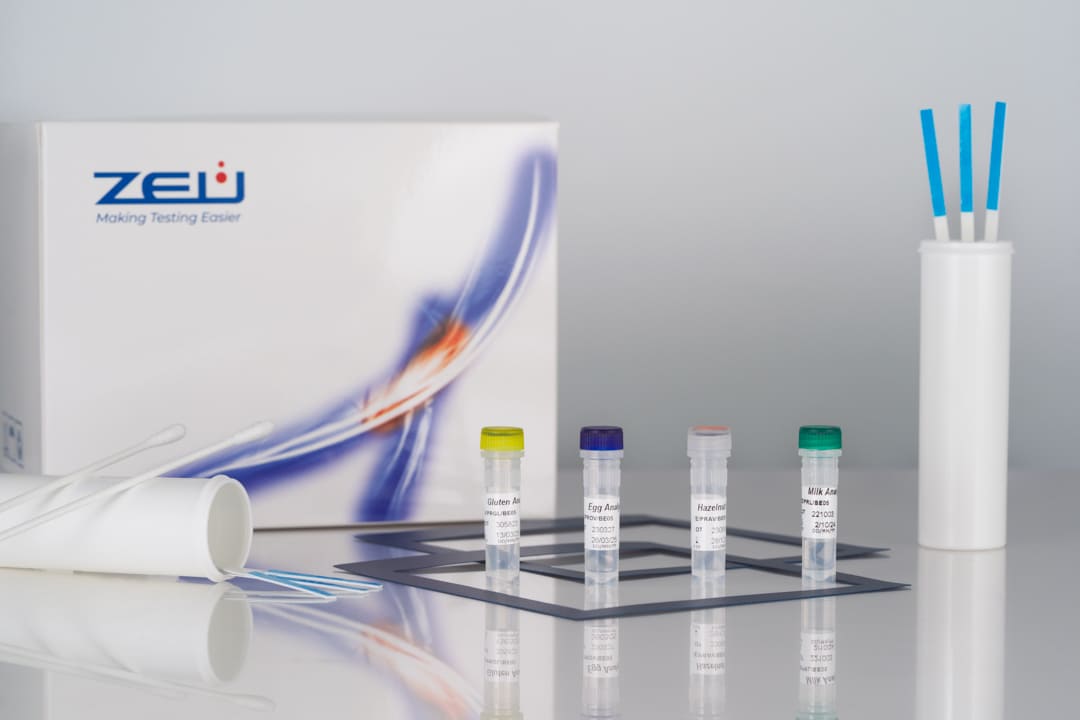
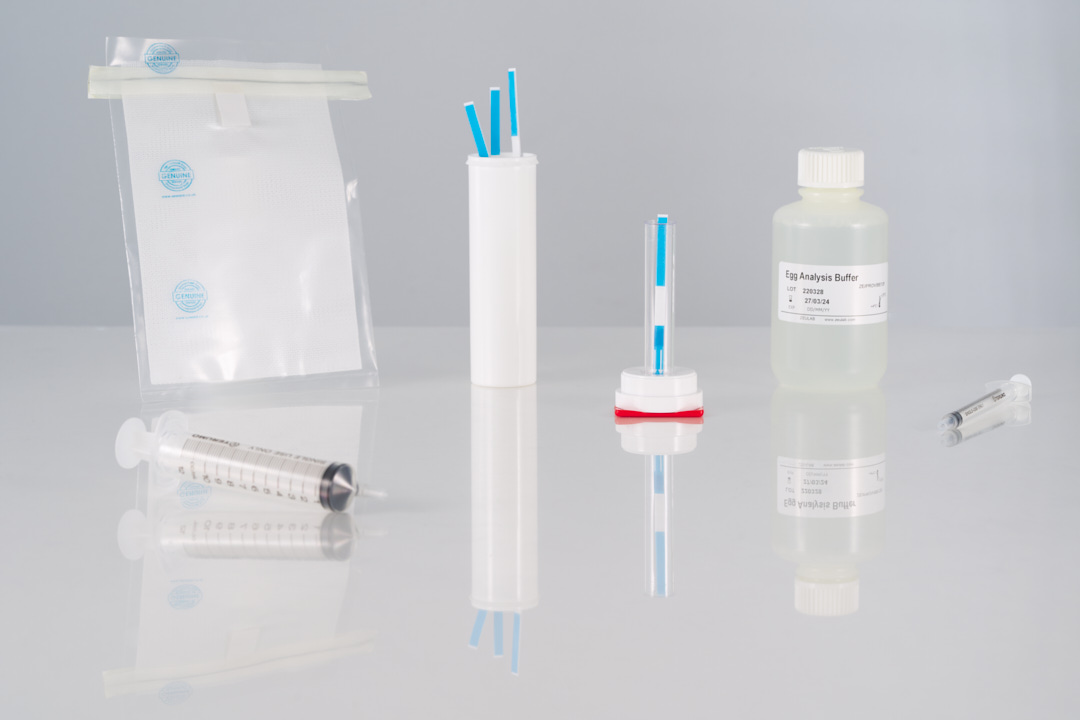
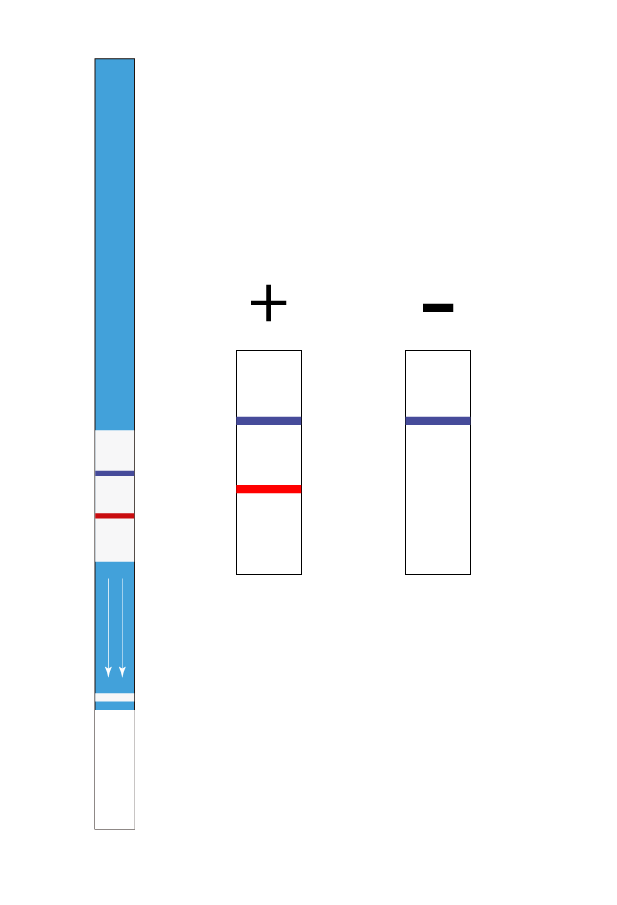- Limit of detection: 0.5 ppm of egg protein.
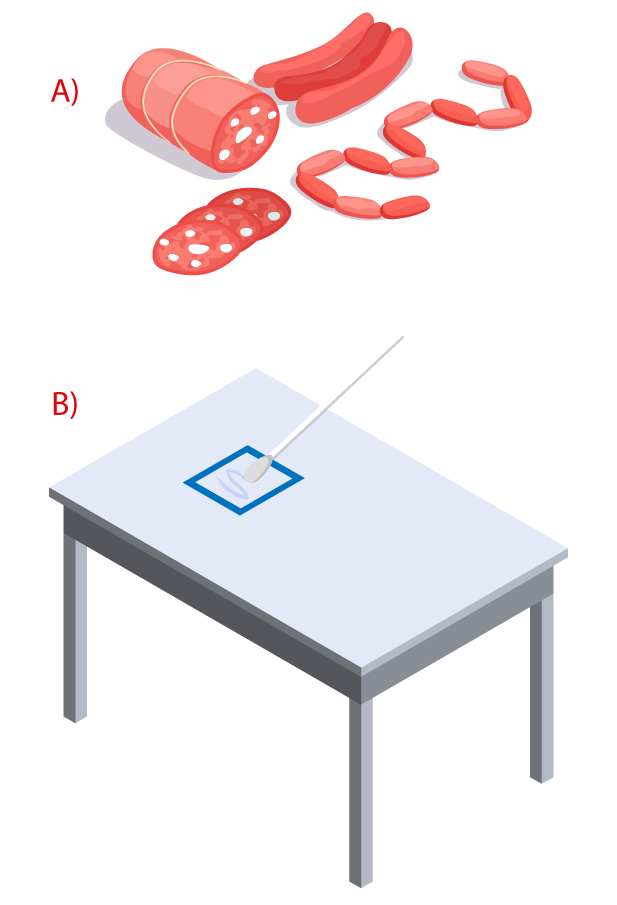
- Limit of detection in surfaces: 0.25 µg of protein.
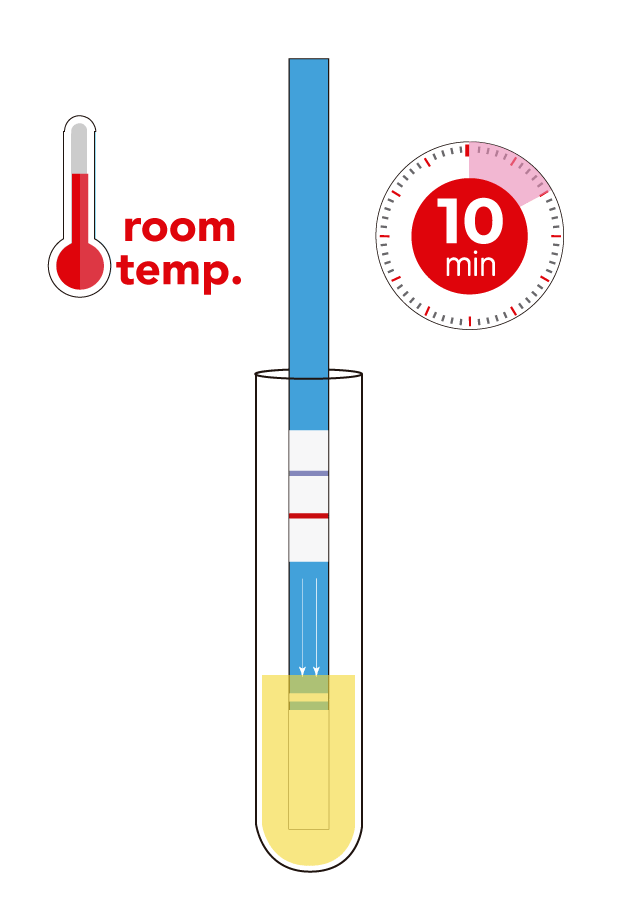
- Assay time: 10 min.
Validations:
Internal validation following International guidelines:
Appendix M: Validation Procedures for Quantitative. Food Allergen ELISA Methods: Community Guidance and Best Practices. AOAC 2012 (http://www.eoma.aoac.org/app_m.pdf).
Appendix F: Guidelines for Standard Method Performance Requirements. Official Methods of Analysis (2016), AOAC INTERNATIONAL, Rockville, MD, USA (http://www.eoma.aoac.org/app_f.pdf)
Standard Method Performance Requirements (SMPRs®) for Quantitation of Chicken Egg by ELISA-Based Methods. AOAC (SMPRs®) 2017.020 (http://members.aoac.org/aoac_prod_imis/AOAC_Docs/ISPAM/SMPR_2017.020.pdf)
Standard Method Performance Requirements (SMPRs®) for Detection and Quantitation of Selected Food Allergens. AOAC SMPR 2016.002. AOAC INTERNATIONAL, Rockville, MD, USA (https://www.aoac.org/aoac_prod_imis/AOAC_Docs/SMPRs/SMPR%202016_002.pdf).
Galan-Malo et al. (2017) Detection of egg and milk residues on working surfaces by ELISA and lateral flow immunoassays test. Food Control 74: 45-53.
Supporting bibliography for validation of antibodies and extraction methods of food samples:
De Luis et al. (2008) Evaluation of indirect competitive and double antibody sandwich ELISA tests to determine β-lactoglobulin and ovomucoid in model processed foods. Food and Agricultural Immunology, 19: 339-350.
Legislation:
Regulation (EU) No 1169/2011 of the European Parliament and of the Council of 25 October 2011 on the provision of food information to consumers, amending Regulations (EC) No 1924/2006 and (EC) No 1925/2006 of the European Parliament and of the Council, and repealing Commission Directive 87/250/EEC, Council Directive 90/496/EEC, Commission Directive 1999/10/EC, Directive 2000/13/EC of the European Parliament and of the Council, Commission Directives 2002/67/EC and 2008/5/EC and Commission Regulation (EC) No 608/2004 Text with EEA relevance.